What Are Ceramics?
Ceramics encompass such a vast array of materials that a concise definition is almost impossible. However, one workable definition of ceramics is a refractory, inorganic, and nonmetallic material. Ceramics can be divided into two classes: traditional and advanced. Traditional ceramics include clay products, silicate glass and cement; while advanced ceramics consist of carbides (SiC), pure oxides (Al2O3), nitrides (Si3N4), non-silicate glasses and many others. Ceramics offer many advantages compared to other materials. They are harder and stiffer than steel; more heat and corrosion resistant than metals or polymers; less dense than most metals and their alloys; and their raw materials are both plentiful and inexpensive. Ceramic materials display a wide range of properties which facilitate their use in many different product areas.
Product Area |
Product |
Aerospace |
space shuttle tiles, thermal barriers, high temperature glass windows, fuel cells |
Consumer Uses |
glassware, windows, pottery, Corning® ware, magnets, dinnerware, ceramic tiles, lenses, home electronics, microwave transducers |
Automotive
|
catalytic converters, ceramic filters, airbag sensors, ceramic rotors, valves, spark plugs, pressure sensors, thermistors, vibration sensors, oxygen sensors, safety glass windshields, piston rings |
Medical (Bioceramics) |
orthopedic joint replacement, prosthesis, dental restoration, bone implants |
Military
|
structural components for ground, air and naval vehicles, missiles, sensors |
Computers |
insulators, resistors, superconductors, capacitors, ferroelectric components, microelectronic packaging |
Other Industries
|
bricks, cement, membranes and filters, lab equipment |
Communications |
fiber optic/laser communications, TV and radio components, microphones |
Humans have found applications for ceramics for the past 30,000 years; every day new and different applications are being discovered. This truly makes ceramics a stone age material, with space age qualities.
Future Trends
Ceramics of the past were mostly of artistic and domestic value. Ceramics of the present have many industrial applications. Imagine what the next generation (your kids) will be doing because of advances in ceramics.
Imagine |
The Future with Ceramics |
Hand-held interactive videos that fit in your pocket |
The electronic field looks ahead to microminiaturization of electronic devices. Ceramic engineers will turn nonfunctional packaging parts into functional components of the device. To accomplish this, new ceramic materials will be developed along with new methods to process them. |
Phones that won’t ring; rings that will be phones with no dial pad |
The communication industry was revolutionized with the development of fiber optics. Along with microminiaturization of components will come the incorporation of opto-electronic integrated circuits. |
A 300 mph train ride into Fantasy Land
|
High temperature superconductors will open the doors to magnetic levitation vehicles, cheap electricity, and improved MRI (magnetic resonance imaging). With micro-applications of superconductors through thin film tapes in sensors and memory storage devices, the use of superconductors will take-off. |
A high speed electric car powered with a fuel cell and full of high tech sensors that practically drive the car for you
|
The automobile industry, which already incorporates seventy pounds of ceramics into a car, is looking to the field of ceramics to provide improved sensors of motion, gas compositions, electrical and thermal changes; as well as light weight, high strength and high temperature components for the engines. For the conservation of energy and environmental protection, ceramics seem to be a viable possibility in the use of ceramic fuel cells, batteries, photovoltaic cells, and fiber optic transmission of energy. |
A best friend that‘s bionic/andromic with microscopic hearing and seeing devices and a skeletal system all made from ceramics |
Besides the ceramic applications in medical diagnostic instruments, the field of bioceramics for bone replacement and chemotherapy release capsules is here. As ceramic materials improve in terms of strength, nonreactivity, compatibility, longevity, porosity for tissue growth, and lower costs, more use of ceramic devices will be seen. |
Scientific Principles
Introduction:
Ceramics have characteristics that enable them to be used in a wide variety of applications including:
• high heat capacity and low heat conductance
• corrosion resistance
• electrically insulating, semiconducting, or superconducting
• nonmagnetic and magnetic
• hard and strong, but brittle
The diversity in their properties stems from their bonding and crystal structures.
Atomic Bonding:
Two types of bonding mechanisms occur in ceramic materials, ionic and covalent. Often these mechanisms co-exist in the same ceramic material. Each type of bond leads to different characteristics.
Ionic bonds most often occur between metallic and nonmetallic elements that have large differences in their electronegativities. Ionically-bonded structures tend to have rather high melting points, since the bonds are strong and non-directional.
The other major bonding mechanism in ceramic structures is the covalent bond. Unlike ionic bonds where electrons are transferred, atoms bonded covalently share electrons. Usually the elements involved are nonmetallic and have small electronegativity differences.
Many ceramic materials contain both ionic and covalent bonding. The overall properties of these materials depend on the dominant bonding mechanism. Compounds that are either mostly ionic or mostly covalent have higher melting points than compounds in which
neither kind of bonding predominates.
Table 1: Comparison of % Covalent and Ionic character with several ceramic compound's melting points.
Ceramic Compound |
Melting Point ˚C |
% Covalent character |
% Ionic character |
Magnesium Oxide |
2798˚ |
27% |
73% |
Aluminum Oxide |
2050˚ |
37% |
63% |
Silicon Dioxide |
1715˚ |
49% |
51% |
Silicon Nitride |
1900˚ |
70% |
30% |
Silicon Carbide |
2500˚ |
89% |
11% |
Classification:
Ceramic materials can be divided into two classes: crystalline and amorphous (noncrystalline). In crystalline materials, a lattice point is occupied either by atoms or ions depending on the bonding mechanism. These atoms (or ions) are arranged in a regularly repeating pattern in three dimensions (i.e., they have long-range order). In contrast, in amorphous materials, the atoms exhibit only short-range order. Some ceramic materials, like silicon dioxide (SiO2), can exist in either form. A crystalline form of SiO2 results when this material is slowly cooled from a temperature (T>TMP @1723˙C). Rapid cooling favors noncrystalline formation since time is not allowed for ordered arrangements to form.
Crystalline Silicon dioxide Amorphous Silicon dioxide
(regular pattern) (random pattern)
Figure 1: Comparison in the physical strucuture of both crystalline and amorphous Silicon dioxide
The type of bonding (ionic or covalent) and the internal structure (crystalline or amorphous) affects the properties of ceramic materials. The mechanical, electrical, thermal, and optical properties of ceramics will be discussed in the following sections.
Thermal Properties:
The most important thermal properties of ceramic materials are heat capacity, thermal expansion coefficient, and thermal conductivity. Many applications of ceramics, such as their use as insulating materials, are related to these properties.
Thermal energy can be either stored or transmitted by a solid. The ability of a material to absorb heat from its surrounding is its heat capacity. In solid materials at T > 0 K, atoms are constantly vibrating. The atomic vibrations are also affected by the vibrations of adjacent atoms through bonding. Hence, vibrations can be transmitted across the solid. The higher the temperature, the higher the frequency of vibration and the shorter the wavelength of the associated elastic deformation.
The potential energy between two bonded atoms can be schematically represented by a diagram:
Figure 2: Graph depicting the potential energy between two bonded atoms
The distance at which there is minimum energy (potential well) represents what is usually described as the bond length. A good analogy is a sphere attached to a spring, with the equilibrium position of the spring corresponding to the atom at the bond length (potential well). When the spring is either compressed or stretched from its equilibrium position, the force pulling it back to the equilibrium position is directly proportional to the displacement (Hooke's law). Once displaced, the frequency of oscillation is greatest when there is a large spring constant and low mass ball. Ceramics generally have strong bonds and light atoms. Thus, they can have high frequency vibrations of the atoms with small disturbances in the crystal lattice. The result is that they typically have both high heat capacities and high melting temperatures.
As temperature increases, the vibrational amplitude of the bonds increases. The asymmetry of the curve shows that the interatomic distance also increases with temperature, and this is observed as thermal expansion. Compared to other materials, ceramics with strong bonds have potential energy curves that are deep and narrow and correspondingly small thermal expansion coefficients.
The conduction of heat through a solid involves the transfer of energy between vibrating atoms. Extending the analogy, consider each sphere (atom) to be connected to its neighbors by a network of springs (bonds). The vibration of each atom affects the motion of neighboring atoms, and the result is elastic waves that propagate through the solid. At low temperatures (up to about 400˚C), energy travels through the material predominantly via phonons, elastic waves that travel at the speed of sound. Phonons are the result of particle vibrations which increase in frequency and amplitude as temperature increases.
Phonons travel through the material until they are scattered, either through phonon-phonon interactions* or at lattice imperfections. Phonon conductivity generally decreases with increasing temperature in crystalline materials as the amount of scattering increases. Amorphous ceramics which lack the ordered lattice undergo even greater scattering, and therefore are poor conductors. Those ceramic materials that are composed of particles of similar size and mass with simple structures (such as diamond or BeO) undergo the smallest amount of scattering and therefore have the greatest conductivity.
At higher temperatures, photon conductivity (radiation) becomes the predominant mechanism of energy transfer. This is a rapid sequence of absorptions and emissions of photons that travel at the speed of light. This mode of conduction is especially important in glass, transparent crystalline ceramics, and porous ceramics. In these materials, thermal conductivity increases with increased temperature.
Although the thermal conductivity is affected by faults or defects in the crystal structure, the insulating properties of ceramics essentially depend on microscopic imperfections. The transmission of either type of wave (phonon or photon) is interrupted by grain boundaries and pores, so that more porous materials are better insulators. The use of ceramic insulating materials to line kilns and industrial furnaces are one application of the insulating properties of ceramic materials.
The electron mechanism of heat transport is relatively unimportant in ceramics because charge is localized. This mechanism is very important, however, in metals which have large numbers of free (delocalized) electrons.
*Phonon-phonon interactions are another consequence of the asymmetry in the interaction potential between atoms. When different phonons overlap at the location of a particular atom, the vibrational amplitudes superimpose. In the asymmetrical potential well, the curvature varies as a function of the displacement. This means that the spring constant by which the atom is retained also changes. Hence the atom has the tendency to vibrate with a different frequency, which produces a different phonon.
Table 2: Comparison of thermal properties of different ceramic materials.
Material |
Melting Temp.(oC) |
Heat Capacity (J/kg.K) |
Coefficient of Linear Expansion 1/oCx10-6 |
Thermal Conductiv-ity (W/m.K) |
Aluminum metal |
660 |
900 |
23.6 |
247 |
Copper metal |
1063 |
386 |
16.5 |
398 |
Alumina |
2050 |
775 |
8.8 |
30.1 |
Fused silica |
1650 |
740 |
0.5 |
2.0 |
Soda-lime glass |
700 |
840 |
9.0 |
1.7 |
Polyethylene |
120 |
2100 |
60-220 |
0.38 |
Polystyrene |
65-75 |
1360 |
50-85 |
0.13 |
One of the most interesting high-temperature applications of ceramic materials is their use on the space shuttle. Almost the entire exterior of the shuttle is covered with ceramic tiles made from high purity amorphous silica fibers. Those exposed to the highest temperatures have an added layer of high-emittance glass. These tiles can tolerate temperatures up to 1480˚ C for a limited amount of time. Some of the high temperatures experienced by the shuttle during entry and ascent are shown in Figure 3.
Figure 3: Diagram of space shuttle's ascent and descent temperatures
The melting point of aluminum is 660˚C. The tiles keep the temperature of the aluminum shell of the shuttle at or below 175˚C while the exterior temperatures can exceed 1400˚ C. The tiles cool off rapidly, so that after exposure to such high temperatures they are cool enough to be held in the bare hand in about 10 seconds. Surprisingly, the thickness of these ceramic tiles varies from only 0.5 inches to 3.5 inches.
Figure 4: Graph of inner temperature of tile versus tile thickness.
The shuttle also uses ceramic applications in fabrics for gap fillers and thermal barriers, reinforced carbon-carbon composites for the nose cone and wing leading edges, and high temperature glass windows.
Ceramic Processing:
Processing of ceramic materials describes the way in which ceramic objects (e.g., glass windows, turbocharger rotor blades, optical fibers, capacitors) are produced.
Processing begins with the raw materials needed to produce the finished components, and includes many individual steps that differ significantly depending on the type of ceramic material, crystalline versus glass.
Processing of Crystalline Ceramics Glass Processing
Raw Material Selection Raw Material Selection
Preparation Melting
Consolidation Pouring
Sintering Annealing
Raw material selection involves obtaining and preparing the right materials for the final product. Traditional ceramics use various forms of clay. Glass makers start with primarily silica. Advanced ceramics use several different raw materials depending on the applications (i.e., properties needed).
Material Uses
Al2O3 (aluminum oxide) Spark-plug insulating bodies, substrates for microelectronic packaging
MgO (magnesium oxide) electrical insulators, refractory brick
SiO2 (Silicon dioxide) cookware, optical fibers
ZrO2 (zirconium oxide) cubic zirconia, oxygen sensors
SiC (silicon carbide) kiln parts, heating elements, abrasives
Si3N4 (silicon nitride) turbocharger rotors, piston valves
For crystalline ceramics, the characteristics of the raw materials (powders) such as their particle size and purity are very important as they affect the structure (e.g., grain size) and properties (e.g., strength) of the final component. Since strength increases with decreasing grain size, most starting powders are milled (or ground) to produce a fine powder (diameter < 1 µm). Since dry powders are difficult to shape, processing additives like water, polymers, etc. are added to improve their plasticity.
Consolidation involves forming the ceramic mixture into the specified shape. There are many techniques available for this step:
Figure 11: Ceramic processing aides.
Sintering is the final step in the process. Sintering at high temperatures (800˚ to 1800˚ C) causes densification that gives the ceramic product its strength and other properties. During this process, the individual ceramic particles coalesce to form a continuous solid network and pores are eliminated. Typically, the mictrostructure of the sintered product contains dense grains, where an individual grain is composed of many starting particles.
Figure 12: Microstructure of raw, formed, and sintered ceramic products
Glass processing is different from crystalline processing. One of the considerations that must be examined is the solidifying behavior of glass. Glasses are most commonly made by rapidly quenching a melt. This means that the elements making up the glass material are unable to move into positions that allow them to form the crystalline regularity. The result is that the glass structure is disordered or amorphous.
One of the most notable characteristics of glasses is the way they change between solid and liquid states. Unlike crystals, which transform abruptly at a precise temperature (i.e., their melting point) glasses undergo a gradual transition. Between the melting temperature (Tm) of a substance and the so-called glass transition temperature (Tg), the substance is considered a supercooled liquid. When glass is worked between Tg and Tm, one can achieve virtually any shape. The glass blowing technique is a fascinating demonstration of the incredible ability to deform a glass.
Figure 13: Specific Volume vs. Temperature graph for a typical ceramic material
Glass processing does not require an optimum size particle (although smaller pieces melt faster). The selections of glass raw materials and chemical additives (which, for example, can alter the color of the glass) are heated up (700˚ - 1600˚ C), melted and finally poured onto or into a quick-cool form or plate. There are four different forming techniques used to fabricate glass.
Technique Application
Pressing Table ware
Blowing Jars
Drawing Windows
Fiber forming Fiber optics
During the glass formation, there may be stresses that have been introduced by rapid cooling or special treatments that the glass needs (such as layering or strengthening). Additional heat treatment is needed to “heal” the glass. Annealing, in which the glass is heated to the annealing point (a temperature just below the softening point where the viscosity is approximately 108 Poise) and then slowly cooled to room temperature, is one such process. Tempering is also a follow-up heat treatment in glass processing in which the glass is reheated and cooled in oil or a jet of air so that the internal and external parts have different properties. The tempering reduces the tendency of glass to fail. Tempered glass can then be used in conditions prone to stresses like car windows.
References
Baker,W. et al., Synthetic Materials: Applications in Biology/Chemistry, Center for Occupational Research and Development, Waco, TX (1993).
Buchanan, R. (editor),"Electronic Ceramics," Ceramic Bulletin, 63:4 (1984) pp. 567-594.
Callister, W. D., Materials Science and Engineering, an Introduction,John Wiley and Sons, NY (1994).
Chandler, M., Ceramics in the Modern World, Double Day & Co. Inc., Garden City, NY (1967).
Ellis, A. B. et al.,Teaching General Chemistry: A Materials Science Companion, American Chemical Society, Washington, D.C. (1993).
Evans, J. & DeJonghe, L.C,The Production of Inorganic Materials, Macmillan Publishing Company, NY (1991).
Halliday, D. & Resnick, R., Physics, John Wiley and Sons, NY (1978).
Hench, L., "Bioceramics: from Concept to Clinic," American Ceramic Society Bulletin, 72:4 (April 1993) pp. 93-98.
Hlavac, J., Technology of Glass and Ceramics, Elsevier Scientific Press, Oxford (1983).
Holscher, H. H., "Hollow and Specialty Glass: Background and Challenge," Owens- Illinois Bulletin, reprinted from The Glass Industry,Vol. 46, Glass Publishing Co., NY (1965).
Hove, J. E. and Riley,W. C., Modern Ceramics, John Wiley and Sons, NY (1965).
Ichinose, Noboru, Introduction to Fine Ceramics, John Wiley and Sons, NY (1987).
Kendall, K., "Ceramics in Fuel Cells," Ceramic Bulletin, 70:7 (1991) pp. 1159-1160.
Ketron, L. A., "Fiber Optics: The Ultimate Communications Media," Ceramic Bulletin, 66:11 (1987) pp. 1571-1578.
Kingery, W. D., Bowen, H. K., Uhlmann, D. R., Introduction to Ceramics, John Wiley and Sons, NY (1976).
Kingery, W. D., The Changing Roles of Ceramics in Society, American Ceramic Society, Westerville, OH (1990).
Korb, L. J., et al., "The Shuttle Orbiter Thermal Protection System," Bulletin American Ceramic Society, 60:11 (1981) pp. 1188-1193.
Lewis, J., "Superconductivity: Conventional vs. High Tc Superconductors," unpublished University of Illinois at Urbana Champaign, MAST workshop.
Mitchell, Lane, Ceramics: Stone Age to Space Age, McGraw-Hill, Inc., NY (1963).
Musicant, Solomon, What Every Engineer Should Know about Ceramics, Marcel Dekker, Inc., NY (1991).
Norton, F. H., Elements of Ceramics, Addison-Wesley, Cambridge, MA (1952).
Orna, M. V., Schreck, J. O., & Heikkinen, H., ChemSource. Vol. 2, ChemSource, Inc. New Rochelle, NY (1994).
Reed, James., Principles of Ceramic Processing, John Wiley and Sons, NY (1988).
Rhodes, D.,Clay and Glazes for the Potter, Clinton Book Co., Radnor, PA (1974).
Richerson, D. W., Modern Ceramic Engineering, Marcel Dekker, Inc., NY (1982).
Scholes, S. R., Modern Glass Practice, Industrial Publications, NY (1952).
Schwartz, M. M. (editor), Engineering Applications of Ceramic Materials, American Society for Metals, Metals Park, OH (1985).
Sheppard, L. M., "Automotive Performance Accelerates with Ceramics," Ceramic Bulletin, 69:6 (1990) pp. 1011-1021.
Sheppard, L. M., "Automotive Sensors Improve Driving Performance," Ceramic Bulletin, 71:6 (1992) pp. 905-912.
Smith,W. F., Foundations of Material Science and Engineering, McGraw Hill, Inc. (1993).
Tipler, P., Physics, Worth Publishers, Inc. (1982).
Viechnicki, D. J., Slavin, M. J., & Kliman, M. I., "Development and Current Status of Armor Ceramics," Ceramic Bulletin, 70:6 (1991) pp. 1035-1039.
Vincenzini, P., Fundamentals of Ceramic Engineering, Elsevier Applied Science, NY (1991).
Weast, R. C. (editor), CRC Handbook of Chemistry and Physics, CRC Press, Inc. Boca Raton, FL (1985).
Wellock and Deckman, Ceramic Bulletin, Vol. 71, No. 1. (1992).
Resources
ALCOA : 1-800-643-8771
American Ceramic Society
735 Ceramic Place
Westerville, OH 43081-8720
614-890-4700
Materials and Equipment Grid
Materials |
Clay Labs |
Glass Labs |
Demonstrations |
Plaster of Paris |
AS |
|
|
Clay Slip |
AS |
|
|
Clay |
AS |
|
|
Beam Forms |
HIS |
|
|
Mass balance |
LE |
|
|
Grad. Cylinder |
|
|
LE |
Acid Solution |
|
|
LE |
Basic Solution |
|
|
LE |
pH Indicator |
|
|
LE |
Kiln |
LE and O |
|
|
3 Pt. apparatus |
LE and O |
|
|
Candle |
LE |
|
|
Roofing Nails |
HIS |
|
|
Burner |
LE |
|
|
Borax |
|
LE |
|
Nichrome wire |
|
LE |
|
Soft Glass Tubing |
|
LE |
|
Glass Cutter |
|
LE |
|
Pyrex rod or tubing |
|
LE |
|
ohmmeter |
|
LE or E |
|
2 Alligator clips |
|
LE or H |
|
Ring stand |
|
LE |
|
Glass Rod |
|
LE |
|
Flash Light |
|
O |
|
Rubber Stopper |
|
LE |
|
LE = Lab equipment |
O=Other |
H=Hardware |
|
GLOSSARY
Abrasive: A hard material used to grind, cut or wear.
Absorption: The inclusion of the energy of a photon within a substance.
Amorphous: A noncrystalline substance, atoms lack long range order.
Annealing: Heat treatment to alter properties.
Annealing point (glass): Temperature at which stresses are removed.
Atomic vibration: Movement of an atom within a substance.
Band gap energy: Energy difference between the valence and conduction bands.
Brittle fracture: A break that occurs by rapid crack propagation.
Capacitance (C): Charge storing capability.
Cement: A material that binds particles together in a mixture.
Ceramic: A compound of metallic and nonmetallic elements.
Color: Wavelengths of light perceived by the eye.
Component: A part, or device.
Conduction band: Carries the excited conduction electrons.
Conductivity: The ability to carry an electric current (electricity) or thermal energy (heat).
Covalent bond: Bonding by sharing electrons.
Crystalline: A solid with a repeating three-dimensional unit cell.
Crystal structure: The orderly arrangement of the atoms or ions within a crystal.
Diamagnetism: Weakly repelled from a magnetic field.
Dielectric: An insulator.
Dielectric constant: Relative electrical permittivity of a material as compared to a perfect vacuum.
Dielectric (breakdown) strength: The amount of electricity needed to start an electric current flow in a dielectric material.
Ductile fracture: Break accompanied by large plastic deformation.
Elastic deformation: Change in shape that returns when a stress is removed.
Elastic Modulus: Ratio of stress to strain in elastic deformation, measure of elasticity.
Electric field: The gradient of voltage.
Electronegativity: The attraction of an atom for shared electrons.
Electron volt (eV): Unit of energy equivalent to the energy gained by an electron when it falls through an electric potential of one volt.
Excited state: An energy state to which an electron may move by the absorption of energy.
Fiber Optics: The technology of transferring information as light pulses through long thin fibers, usually made of glass.
Firing: High temperature processing to increase densification in a product.
Fluorescence: Light that is emitted a short period of time after an electron has been excited.
Fracture toughness (Kc): Measure of a material's resistance to crack propagation.
Glass: An amorphous solid showing characteristic specific volume behavior over a certain temperature range.
Glass - ceramic: Crystalline ceramic material that was formed by heat treating glass.
Glass transition temperature (Tg): Temperature at which a glass changes from a
supercooled liquid into a solid.
Grain: Individual crystal in a polycrystalline material.
Grain boundary: The boundary between grains (or crystals) that are misoriented with respect to one another.
Green ceramic body: Ceramic object that is dried but not fired.
Ground state: Lowest electron energy state.
Hardness: Resistance to deformation.
Heat capacity: Heat required to produce a unit increase in temperature per quantity of material.
Imperfection: Flaw, any deviation from perfection, as in a crystal.
Index of refraction: Ratio of the speed of light in a vacuum to the speed of light in a medium.
Insulator: Material that does not conduct electricity (electrical) or heat (thermal).
Ionic bond: Electrostatic force between oppositely charged ions.
Laser: Source of coherent light (Light Amplification by Stimulated Emission of Radiation).
Lattice: The regular arrangement of points in a crystal.
Luminescence: Emission of visible light when an electron returns to the ground state from an excited state.
Magnetic field strength: Intensity of an applied magnetic field.
Microstructure: Structural features that can be observed with a microscope.
Noncrystalline: Amorphous, with no long-range atomic order.
Opaque: Material that does not transmit light.
Phonon: Quantum of vibrational energy.
Phosphorescence: Luminescence that lasts for more than one second.
Photovoltaic cells: A device capable of converting light energy to electricity.
Photoconductivity: Electrical conductivity induced by light.
Photon: Quantum of electromagnetic energy.
Piezoelectric: Material that produces an electrical response to a mechanical force.
Plastic deformation: Permanent deformation, change of shape.
Polycrystalline: Composed of more than one crystal or grain.
Porcelain: A durable ceramic composite made by firing clay, feldspar and quartz together.
Reflection: Deflection of light at the interface between two materials.
Refraction: Bending of light as it passes from one medium into another.
Refractory: Material that can be exposed to high temperature without deterioration.
Resistivity: Measure of resistance to passage of electrical current (reciprocal of conductivity).
Semiconductor: Nonmetallic material that has a relatively narrow energy band gap.
Sintering: Coalescence of individual ceramic particles into a continuous solid phase at a high temperature.
Slip: Mixture of clay with water that can be poured into a mold.
Slip casting: Method of making ceramic objects by pouring slip into a mold.
Softening point (glass): Maximum temperature a glass can be heated before it permanently deforms.
Smart materials: Materials able to detect a change in the environment and react to it.
Specific volume: Volume per unit mass, reciprocal of density.
Strain: Change in length of a sample in the direction of an applied stress.
Stress: Force applied to a sample divided by its cross-sectional area.
Structural clay products: Ceramic objects made mainly of clay and used in structural applications.
Structure: Arrangement of internal components.
Superconductivity: Disappearance of electrical resistivity at low temperatures.
Supercooling: Cooling below the normal temperature for a phase change, without the change occurring.
Tensile strength: Maximum stress without fracture.
Thermal expansion coefficient, linear: Fractional change in length divided by change in temperature, a measure of a materials tendency to expand when heated.
Thermal stress: Residual stress caused by a change in temperature.
Thermal tempering: The introduction of residual compressive stresses to increase the strength of glass.
Toughness: Energy absorbed by a material as it fractures, a measure of its resistance to fracture.
Transgranular fracture: Fracture by crack propagation through the grains.
Translucent: Transmits light diffusely.
Transparent: Transmits light clearly.
Unit cell: The basic repeating unit in a crystal.
Whiteware: Clay-based ceramic that turns white after firing.
Source: http://matse1.matse.illinois.edu/ceramics/ceramics.doc
Web site to visit: http://matse1.matse.illinois.edu/
Author of the text: indicated on the source document of the above text
Combinations of clay, feldspar and silica (cheap)
Nearly pure compounds of
![]() oxides, carbides or nitrides (expensive)
oxides, carbides or nitrides (expensive)
Non-silicate oxide ceramics Non-oxide ceramics
-Al203 Alumina -Si3N4 Silicon nitride
-ZrO2 Zirconia -SiC Silicon carbide
Structural Clay Products Whitewares
brick more carefully controlled compositions
-tile -pottery
-sewer pipe -porcelain
-cement -sanitary ware
Clays
Whitewares and Structural Clay Products
Refractories
Abrasives
Cements
Note: Concrete is a composite material made up of cement, large aggregates (gravel), and small aggregates (sand). The cement in concrete is similar to the glassy phase in ceramics. It bonds the other phases together.
Portland cement
Ceramic Crystalline Structures

Crystal structures of ionic compounds tend to maximize packing efficiency. (This lowers the overall energy.) The limitations to dense packing are the radius ratio and the need to maintain charge neutrality. The radius ratio can help to predict the CN (and hence the crystal structure.)
If the bonding has some covalent character, then the packing will be less efficient. You can determine the amount of ionic character in a bond by knowing the electronegativities, XA & XB, of the elements in the bond and the following equation: % ionic character = [1 – e -1/4(XA-XB)] x 100%
A tetrahedral position is an interstitial site that if occupied, would have 4 neighbors.
An octahedral position is an interstitial site that if occupied, would have 6 neighbors.
Where are the octahedral and tetrahedral sites in the fcc crystal structure?
Where are the octahedral and tetrahedral sites in the hcp crystal structure?
Some relatively simple ceramic structures:
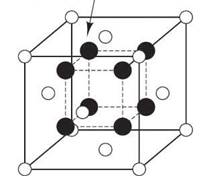
Silica (SiO2) has half ionic and half covalent character. (The percent of ionic character is about 50% as calculated from the above equation.) A CN of 4 is predicted for the radius ratio of 0.29 for ionic compounds. A bond angle of 105o is predicted for covalent bonding. Silica is allotropic with at least five different crystalline forms depending on temperature and pressure conditions. However, each form has every corner of an SiO4 tetrahedra (every oxygen atom) shared. The sharing of oxygen atoms between the tetrahedra give the overall formula of SiO2. The structures are not dense and have high melting temperatures.
Silicates (or silicate ceramics) are Silica (SiO2) based ceramic clays that contain SiO44- tetrahedra in various arrangements. Many naturally occurring minerals such as clays, feldspars and micas are silicates. Many ceramic materials contain silicate structures. They include glass, cement, brick, electrical insulative materials, etc.
The general nature of silicate structures is the connection of the SiO44- tetrahedra. Additional oxides tend to break up the continuity of these tetrahedra. The remaining connectedness may be in the form of islands, chains or sheets:
Electrical Properties of Ceramics
Ceramics are used in many electrical applications as:
Insulators
prevents the flow of charge or heat. Ceramics make good insulators because the ionic and covalent bonding restricts electron and ion mobility.
Capacitors
store electrical energy by virtue of separating oppositely charged plates with an insulator (also called a dielectric) in between. An example of a ceramic material used as the dielectric for a capacitor is Barium Titanate, Ba2TiO3
Some material properties of insulators (also called dielectrics):
Some examples of ceramic insulators are
Semiconductors
have an intermediate value of electrical conductivity. The most commonly used ceramic semiconducting materials are sintered oxides of Mn, Ni, Fe, Co, and Cu. By varying the amounts of these oxides in the material the desired value of the electrical conductivity can be obtained. These are used for NTC thermistors. A Negative Temperature Coefficient Thermistor is a device that measures temperature by a change in the resistance of the material. As temperature increases, resistance decreases.
Ferroelectrics
have behavior similar to a ferromagnetic material. The relationship between the electric dipole moment density (Polarization, P) and the electric field strength (E) looks exactly like the relationship between magnetic dipole moment density (Magnetization, M) and magnetic field strength (H).
Piezoelectrics
have the distinct property whereby an applied stress induces an electrical voltage. The reverse piezoelectric effect is the phenomena whereby an applied voltage across the material results in a dimensional change. BaTiO3
Mechanical Properties of Ceramic Materials
Ceramics are strong
However, their principle drawback is a disposition to catastrophic fracture in a brittle mode. Hence, the effort in research of these materials is to make them tougher. An example of an advancement in this area is the transformation toughening of partially stabilized Zirconia (PSZ).
Fine grain size ceramics are stronger than coarse grain size ceramics
The reason is different than for metals: If fracture does not initiate at a crack, flaw, pore or inclusion, it will originate at a flaw in a grain boundary. Finer grain size ceramics will have smaller flaw at the grain boundaries. Hence, the finer grain ceramics will be stronger.
The Flexural Strength is an alternate property to Tensile Strength for ceramics
The flexural strength (or modulus of rupture, fracture strength, bend strength) is the (calculated) tensile stress at failure of a specimen subjected to a three or four point load. This is an easier test to carry out for brittle materials than the traditional standard Tensile Test.
Ceramics are brittle
Ceramics have very low toughness values and also fracture toughness values. Recall that brittle fracture consists of formation and propagation of cracks through the cross section in a direction perpendicular to the applied load. Usually the crack growth is transgranular and along specific crystallographic directions.
The reasons that ceramics are so brittle:
Ceramics are much stronger in compression than in tension.
Why?
Some ceramics are subject to Static Fatigue or Delayed Fracture
This is when ceramics fail by slow crack propagation under a static load. The mechanism is a stress-corrosion process at a crack tip.
Ceramics rarely have any sort of fatigue failure
This is due to the lack of plasticity in ceramics; however, there have been recent advances in compressive-compressive stress cycling of polycrystalline alumina.
Ceramics have high hardness values
This makes them excellent abrasive materials used for cutting, grinding and polishing. Alumnia (aluminum oxide, Al2O3) and silicon carbide, SiC are two of the most commonly used manufactured ceramic abrasives. Improvements on these are made by combining zirconium oxide with aluminum oxide. Another important ceramic abrasive is cubic boron nitride (Bortazon).
Ceramics have high melting temperatures
This makes them excellent materials to be used for high temperature applications. Materials used at high temperatures are call refractory materials or refractories. However, creep will occur at these temperatures and so one must be aware of the material’s creep behavior.
Porosity has an adverse affect on a ceramic’s properties.
The modulus of elasticity and the flexural strength will both decrease with porosity.
There is much scatter in the data measuring the properties of ceramic materials
The reason is because much of the data depends on flaw density and size. This, in turn, will depend on fabrication technique, processing and size of the specimen. Hence, the scatter.
silica – (aka flint or quartz) SiO2 is a polymorphic compound. (quartz, tridymite and cristobalite are some of its forms.) It acts as the refractory component of traditional ceramics.
Source: https://fog.ccsf.edu/~wkaufmyn/ENGN45/Course%20Handouts/Chap12_Ceramics.doc
Web site to visit: https://fog.ccsf.edu/
Author of the text: indicated on the source document of the above text
If you are the author of the text above and you not agree to share your knowledge for teaching, research, scholarship (for fair use as indicated in the United States copyrigh low) please send us an e-mail and we will remove your text quickly. Fair use is a limitation and exception to the exclusive right granted by copyright law to the author of a creative work. In United States copyright law, fair use is a doctrine that permits limited use of copyrighted material without acquiring permission from the rights holders. Examples of fair use include commentary, search engines, criticism, news reporting, research, teaching, library archiving and scholarship. It provides for the legal, unlicensed citation or incorporation of copyrighted material in another author's work under a four-factor balancing test. (source: http://en.wikipedia.org/wiki/Fair_use)
The information of medicine and health contained in the site are of a general nature and purpose which is purely informative and for this reason may not replace in any case, the council of a doctor or a qualified entity legally to the profession.
The texts are the property of their respective authors and we thank them for giving us the opportunity to share for free to students, teachers and users of the Web their texts will used only for illustrative educational and scientific purposes only.
All the information in our site are given for nonprofit educational purposes